N2 (g) + 3H2 (g) ⇔ 2NH3 (g) + Energy
Complete the table below by filling in "right" or "left" for the equilibrium shift and "increases" or "decreases" for the concentration of reactants and products.
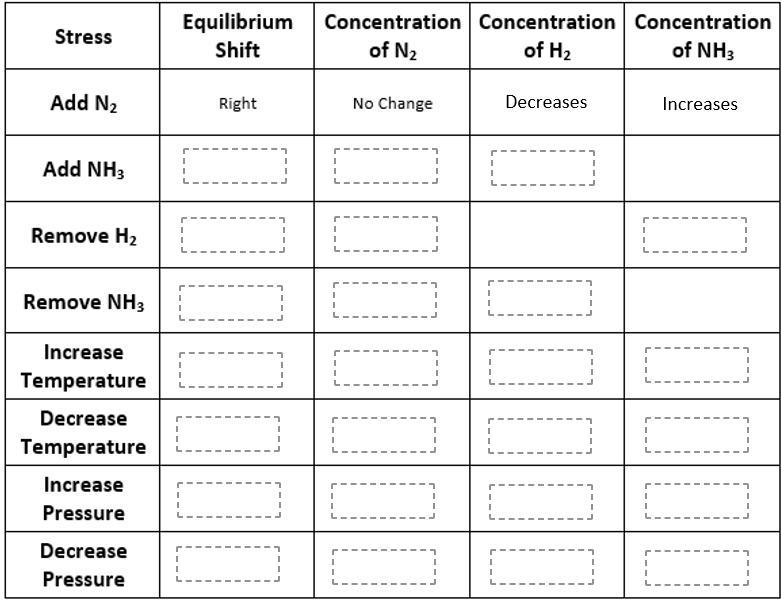
Answers
Answer:
b is the answer
Related Questions
What three things does force cause?
Answers
Forces can cause objects to accelerate , decelerate , stop , start moving , change direction , change shape , or even turn
Select the example of symbiosis, chosen from the list below that is best described by the statement shown.
Answers
Answer:
can you show us the list
Explanation:
Two substances are combined and react. After 10 minutes, the chemical reaction has reached equilibrium. Which values would be equal to zero for the solution?
Answers
The extra conversion of concentration of reactant and product should be zero in order to attaining equlibrium state.
What is equilibrium?Chemical equilibrium refers to the state in which both the reactants and products are present in equal concentrations or amount. In equlibrium, same amount of reactant is converted into product and product into reactant.
So we can conclude that the extra conversion of concentration of reactant and product should be zero in order to attaining equlibrium state.
Learn more about equilibrium here: https://brainly.com/question/517289
why electron configuration is important
Answers
Answer:
So that we know how many electrons are thre in which shell and how many shells are present in an atom....
Explanation:
Hope it helps!!!
Why agar is used as a solidifying agent in culture media instead of gelatin
Answers
Agar is used as a solidifying agent in culture media instead of gelatin because it readily melts and it contains no nutrition for bacteria cultures.
What is agar?Agar is a gelatinous material obtained from red algae and used in the preparation of culture medium.
Agar is used to solidify the culture medium for bacteria growth, although, gelatin can also be used.
However, agar is preferred as a solidifying agent in culture media instead of gelatin because it readily melts and it contains no nutrition for bacteria cultures.
Learn more about agar at: https://brainly.com/question/24554352
A world with both liquid water on its surface and carbon dioxide in its atmosphere is unstable because.
Answers
word equation for water
Answers
Answer:
The actual reaction to make water is a bit more complicated: 2H2 + O2 = 2H2O + Energy. In English, the equation says: To produce two molecules of water (H2O), two molecules of diatomic hydrogen (H2) must be combined with one molecule of diatomic oxygen (O2). Energy will be released in the process.
Explanation:
What is true of a sample of gas as temperature is increased? (3 points)
Answers
As temperature increases, the average kinetic energy of a sample of gas molecules decreases.
Cyanide and water react in a proton transfer reaction to form hydrogen cyanide and hydroxide
Answers
The proton transfer reaction between Cyanide and water can be written as; X^- + H2O -----> HX + OH^-
What is a proton transfer reaction?A proton transfer reaction is one in which a proton is moved from one chemical specie to another.It is in fact and acid - base reaction in the Brownstead - Lowry sense.
The proton transfer reaction between Cyanide and water can be written as(Let the cyanide ion be shown as X);
X^- + H2O -----> HX + OH^-
Learn more about proton transfer: https://brainly.com/question/861100?
The proton transfer reaction to form hydrogen cyanide and hydroxide is
X⁻ + H₂O → HX + OH⁻
What is a proton transfer reaction?A reaction in which a proton H⁻ is removed from one species and accepted by another species.
An example of a proton transfer is Brønsted acid reacting with a Brønsted base.
Thus, the proton transfer reaction to form hydrogen cyanide and hydroxide is X⁻ + H₂O → HX + OH⁻
Learn more about proton transfer
https://brainly.com/question/861100
#SPJ4
Explain in words what is happening in each chemical equation.
nh3 + o2 -> no2- + 3h+ + 2e-
no2- + h2o -> no3- + 2h+ +2e-
Answers
Answer:
Ammonia and oxygen without catalyst | NH3 + O2 → N2 + H2O. With supply of heat, ammonia reacts with oxygen and produce nitrogen gas and water as products. Nitrogen of ammonia is oxidized to nitrogen gas from -3 oxidation state to 0 oxidation state.
Explanation:
find the oxidation state of c in (NH4)2 CO3
Answers
Answer:
Ammonium carbonate is a neutral molecule. Charge on NH4 is +1 , so on (NH4)2 is +2 ( +1 * 2) ,lets take the charge on C to be x and O is -2 , so on O3 is -6 (3*-2). Therefore x= + 4. Hope it helps.
Explanation:
A compound has a percent composition of 38.7% carbon, 9.76% hydrogen, 51.5% oxygen.
Laboratory data shows that the compound's molar mass is 62.0 g/mol. What is the
molecular formula of the compound?
Answers
The molecular formula of the compound that has a percent composition of 38.7% carbon, 9.76% hydrogen, 51.5% oxygen is C2H6O2.
How to calculate molecular formula?The molecular formula can be calculated from the empirical formula. The empirical formula of the compound is calculated as follows:
C = 38.7% = 38.7gH = 9.76% = 9.76gO = 51.5% = 51.5gNext, we convert the mass to moles by dividing by their atomic mass:
C = 38.7 ÷ 12 = 3.23molH = 9.76 ÷ 1 = 9.76molO = 51.5÷ 16 = 3.22molNext, we divide by the smallest (3.22)
C = 1H = 3O = 1Hence, the empirical formula of the compound is CH3O
If the molar mass of the compound is 62g/mol;
(CH3O)n = 62
31n = 62
n = 2
(CH3O)2 = C2H6O2
Therefore, the molecular formula of the compound that has a percent composition of 38.7% carbon, 9.76% hydrogen, 51.5% oxygen is C2H6O2.
Learn more about molecular formula at: https://brainly.com/question/14425592
How many molecules make up 32 g of Oz?
Answers
Answer:
Chemical formula of Oxygen is O2.
We know ,
1 mole of a substance contains 6.022×10^23, Avogadro’s Constat.
Here, Molar mass of O = 16u
And molar mass of O2 = 32u
And 32g of O2 = 1 mole of O2
It means that,
32g of O2 = 6.022×10^23 molecules
Answer:
The correct answer is given in the picture
This ANSWER IS ONLY CORRECT WHEN Oz STAND FOR OZONE
(1.) Once we confirm other planetary systems, what do you think the
next stage of exploration should be?
Answers
The next stage of exploration should be gathering information about the planetary system.
What is the next stage of exploration?Once we confirm other planetary systems, the next stage of exploration should be the use of instruments that helps us to collect information about that planetary objects so we can conclude that the next stage of exploration should be gathering information about the planetary system.
Learn more about exploration here: https://brainly.com/question/13335713
guys can you help me to fill in the missing blanks
Answers
Answer:
Silver NitratePotassium HydroxideAmmonium ChlorideCalcium CarbonateCopper NitrateAmmonium HydroxideAmmonium NitrateSodium SulfateAn unknown gas with a mass of 205 g occupies a volume of 20.0 L at 273 K and 1.00 atm. What is the molar mass of this compound?
Answers
The molar mass of this compound is 0.004 m/g
Firstly, molar mass is moles/grams so we need to solve for the number of moles and we were already given the grams (205 g).
So to solve for moles we were given 205 g, 20.0 L, 273 K and 1.00 atm. Knowing these numbers we can plug them into the ideal gas law, PV=nRT
where P is pressure, V is volume, n is number of moles, R is the gas constant (in this case it is 0.08206) and T is the temperature.
[tex]PV=nRT\\\\n=\frac{PV}{RT}\\\\n=\frac{1.00*20.0}{0.08206*273} =0.893[/tex]
Next, we use moles and grams to solve for molar mass:
[tex]\frac{0.893}{205} =0.004[/tex]
When the carbonator adds CO 2 to the solution, a ________ is added to the reaction, which pushes the reaction in the
________ direction.
a. product, forward
b. product, reverse
c. reactant, forward
d. reactant, reverse
(CO2 is added to H2CO3)
Answers
When the product is added to the solution, it will shift the reaction forward.
What is Carbonation?Carbonation is a form of chemical reaction in which carbon dioxide is added to a solution to give carbonates, bicarbonates, and carbonic acid. In chemistry, the term is used in place of carboxylation to form carboxylic acids
When a product is added to a system, the equilibrium will shift to the left, so as to produce more reactants but if the reactants is remove from the system, equilibrium will also be shifted to the left.
Therefore, When the product is added to the solution, it will shift the reaction forward.
Learn more about carbonation here.
https://brainly.com/question/24848856
100 points
In an equilibrium expression, the concentrations of ___ are on top.
A:products B:solids C:reactants D:liquids
Answers
Answer:
products, it's answer A.
Answer:
products
Explanation:
Look at the diagram. What is substance X?
Enter your answer
What is substance x??
Answers
Molten cryolite is the substance which serves as an electrolyte in the diagram.
What is an Electrolyte?This is defined as a substance that conducts electricity when dissolved in water.
Molten cryolite serves as the electrolyte as it helps dissolve molten aluminium oxide and increases the conductivity of the solution which was why it was chosen as the most appropriate choice.
Read more about Electrolyte here https://brainly.com/question/17089766
Answer: Molten cryolite
Explanation: Molten cryolite serves as the electrolyte as it helps dissolve molten aluminium oxide and increases the conductivity of the solution which was why it was chosen as the most appropriate choice.
Draw the structures of the aldol addition and condensation products of cyclopentanone
Answers
The term aldol condensation has to do with a reation in which a nucleophile attacks the carbonyl group of an aldehyde.
What is alodol condensation?The term aldol condensation has to do with a reation in which a nucleophile attacks the carbonyl group of an aldehyde or a ketone to convert it to the enolate from which attacks another aldehyde or ketone at the carbonyl carbon to form the required product.
The structure of the organic product from the aldol condensation involving cyclopentanone is shown in the image attached to this answer.
Learn more about aldol condensation: https://brainly.com/question/9415260?:
Predict which one of the following salts is used for softening water?
A) [tex] Na_{2} SO_{4}[/tex]
B) [tex] Na_{2} Si O_{3}[/tex]
C) [tex] Na_{2} CO_{3}[/tex] x [tex] 10 H_{2}O[/tex]
D) [tex] NaClO_{3} [/tex]
Answers
Salts such as [tex] Na_{2} SO_{4}[/tex] are used in softening of hard water.
What is hardness of water?Hardness of water is the presence on water of dissolved metallic ions such as calcium and magnesium ions results in the water having additional properties such as taste.
Hardness of water is of significance in the laundry industry as it causes water to lather with difficulty and also produces sc_um.
To remove hardness of water or to soften water, salts such as [tex] Na_{2} SO_{4}[/tex] are used.
Learn more about hardness of water at: https://brainly.com/question/6946622
Calculate the number of atoms in 2.1g of nitrogen
Answers
Answer:
2.1×6.02×1023÷ 14
9.03×1022 atom of nitrogen
Explanation:
A sample of carbon dioxide gas at 125°c and 248 torr occupies a volume of 275 l. What will the gas pressure be if the volume is increased to 321 l at 125°c?.
Answers
The new pressure of gas when the volume of the gas is increase to 321 L at 125 °C is 212.46 torr
Data obtained from the question Initial pressure (P₁) = 248 torrInitial volume (V₁) = 275 LTemperature = 125 °C = constant New Volume (V₂) = 321 LNew pressure (P₂) =?How to determine the new pressure
The new pressure of the gas can be obtained by using the Boyle's law equation since the temperature is constant. This is illustrated below:
P₁V₁ = P₂V₂
248 × 275 = P₂ × 321
68200 = P₂ × 321
Divide both side by 321
P₂ = 68200 / 321
P₂ = 212.46 torr
Learn more about gas laws:
https://brainly.com/question/6844441
How many atoms are there in 12 moles of sulfur
Answers
HELP
Determine the empirical formula of a compound containing 48.38
grams of carbon, 6.74 grams of hydrogen, and 53.5 grams of oxygen.
In an experiment, the molar mass of the compound was determined to be 180.15 g/mol.
What is the molecular formula of the compound?
For both questions, show your work or explain how you determined the formulas by
giving specific values used in calculations.
Answers
Answer:
I recently answered this question. The response I submitted is included below. I beleive my answer should be correct.
Explanation:
Question 1:
C: 48.38g(1mol/12g) = 4.0317
H: 8.12g(1mol/1.01g) = 8.12
O: 53.5g(1mol/16g) = 3.34375
Divide by the smallest amount (3.34375)
C = 4.0317/3.34375 = 1.206 = 1
H = 8.12/3.34375 = 2.42 = 2
O = 3.34375/3.34375 = 1
Empirical formula = CH2O
Question 2:
Molecular formula = n(empirical formula)
n = molar mass (compound)/molar mass (empirical)
Empirical formula: CH2O
Molar mass of CH2O = 12 + 2x1 + 16 = 30 g/mol
Molar mass of compound: 180.15 g/mol
[tex]n = \frac{180.15g/mol}{30g/mol} = 6[/tex]
Molecular formula = C6H12O6
my teacher hasnt graded yet, but i got the same answer as the guy above. i did my best to explain the process for y'all if you genuinely don't understand/know how to do it. THE PROCESS IS VERY LONG, but youll get it eventually if i didnt help lol GOODLUCK
and yes, the molecular formula is C6H12O6
The number of protons and neutrons in an atom is that atoms __ number.
Answers
If you look at the periodic table, you can simply find the mass number either on the top or bottom of the element.
What does the spheres represent?
Please help?
Answers
Answer:
the N at the top is north bottom is south w on your left is west and e of your right is east
What is the main chemical component present in striking surface of a matchbox?
A. Potassium chlorate
B. Phosphorus
C. Potassium
D. Graphite
Answers
Answer:
Phosphorus
Explanation:
Actually that's red Phosphorus
The matchstick contains potassium chlorate at topOn reaction between these when we slides them white Phosphorus is formed which burns the match sticksAnswer:
Option B. ( Phosphorus)
Explanation:
Here, we want to select the option which is the main component present in the striking surface of a matchbox
The correct choice here is phosphorus
Red phosphorus which is an allotrope of phosphorus finds use in this particular case. It is the main chemical component present in the striking surface of a matchbox.
Hope this helps!
If you have any queries please ask.
Compare the relative strength of the two forces A and B. Explain how you determined this comparison by identifying the forces.
Answers
The intermolecular forces present in B will be much stronger than the intermolecular forces present in A.
What are intermolecular forces?The term intermolecular forces has to do with the forces that hold molecules of a substance together in a particular state of matter. Now, we can see that B must contain dipole - dipole interaction while A has none of these.
It then follows that the intermolecular forces present in B will be much stronger than the intermolecular forces present in A.
Learn more about intermolecular forces: https://brainly.com/question/9007693
typical "lead" pencil is actually made from carbon. if the carbon in the pencil weighs 1.50 grams, the pencil has [atoms] atoms of carbon.